










How does corrosion work? -

If two surfaces are rubbing against each other then any corrosion can get worse as the powder produced by small movements can be more abrasive than the original material -






If the structure of a metal is modified, for example because of the heat from welding, the properties of that metal can change. If chromium is removed at metal crystal boundaries, by the formation of chromium carbide for example, then those boundaries will no longer be rustproof and the metal can start to come apart at the surface. >

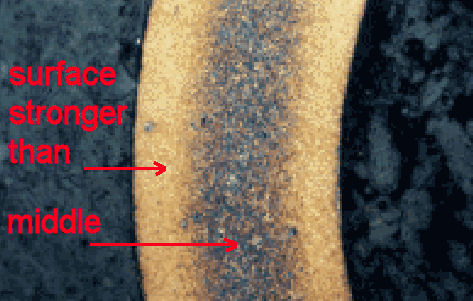
Sometimes, corrosion can strengthen a metal. So if the surroundings are hot and there's carbon available, a hot CO2 atmosphere for example, some of the carbon can enter the metal and form strengthening carbides. The depth of penetration will vary but there will be most carbon at the surface. The surface will be stronger compared to the centre, making the whole component able to carry more load. Formation of nitrides from nitrogen acts similarly. <

There's a special effect that can happen at gaps between components, especially joints and interfaces. If oxide grows on a steel brace holding stone together, or on steel concrete reinforcing bars, then the tensile stresses from the growing oxide can break the stone or concrete.
Stone cracking at the Taj Mahal, India: Concrete cracking away from a building:

If the surfaces form part of a bolted joint, and are oxiding in very hot CO2 for example, the thickness of oxide formed can be greater than the thickness of metal consumed. The oxide then pushes the plates apart and can break the bolt.
Fortunately, if the bolt is strong, then the increase in oxide thickness compresses the plates and washers rather than extending the bolt, so the bolt doesn't break.
2 Aug 2016